42 does potassium conduct electricity
geothermal energy | Description, Uses, History, & Pros and Cons geothermal energy, form of energy conversion in which heat energy from within Earth is captured and harnessed for cooking, bathing, space heating, electrical power generation, and other uses. Heat from Earth's interior generates surface phenomena such as lava flows, geysers, fumaroles, hot springs, and mud pots. The heat is produced mainly by the radioactive decay of potassium, thorium, and ... What Are the Freezing, Melting, and Boiling Points of ... - Owlcation Melting Point/Freezing Point. The terms melting point or freezing point are often interchanged depending on whether a substance is being heated or cooled. For liquids it is known as the freezing point and for solids it is called the melting point. The melting point of a solid and the freezing point of the liquid are normally the same.
Metals and Non-Metals Revision Notes - Collegedunia Metals [Click Here for Sample Questions] The elements that conduct electricity and heat are metals. They are malleable and ductile. Some examples of metals are Iron (Fe), Aluminium (Al), Silver (Ag), Copper (Cu), Gold (Au), Platinum (Pt), Lead (Pb), Potassium (K), Sodium (Na), Calcium (Ca) and Magnesium (Mg), etc. Metals generally have 1 to 3 electrons in the outermost shell (or valency cell ...

Does potassium conduct electricity
Did ammonia conduct electricity? - askingforanswer.com They consist of a metal Mg (magnesium) and a polyatomic ion (sulfate) as well as potassium metal and sulfate ion. Both of these compounds can conduct electricity. Is calcium hydroxide a good conductor of electricity? Calcium Hydroxide can conduct electricity only when it is in aqueous solution state or molten state. Difference Between Strong Base and Weak Base They are also good electrolytes. Strong bases are a good conductor of electricity because the ability to conduct electricity depends on the number of ions present and bases have a concentration of ions.. Some of the examples of strong bases are- Sodium hydroxide (NaOH), Potassium hydroxide (KOH), Lithium hydroxide (LiOH), Calcium hydroxide (Ca(OH)2), Rubidium hydroxide (RbOH), Barium hydroxide ... Electrolyte Challenge: Orange Juice Vs. Sports Drink - Science Buddies The ions in the solution give it the capacity to conduct electricity. Electrolytes, such as sodium and potassium, are present in sweat. Chloride, calcium, and phosphate ions are also electrolytes. The proper concentration of electrolytes in your blood is essential to your health.
Does potassium conduct electricity. Radioactive Wastes From Coal-fired Power Plants | US EPA Radiation Facts. The process of burning coal at coal-fired power plants, called combustion, creates wastes that contain small amounts of naturally-occurring radioactive material. Coal is a fossil fuel used to produce power in the United States. Coal contains trace amounts of naturally-occurring radioactive elements. Properties of metals, metalloids and nonmetals - Wikipedia Sodium, potassium, rubidium, caesium, barium, platinum, gold The common notions that "alkali metal ions (group 1A) always have a +1 charge" [136] and that "transition elements do not form anions" [137] are textbook errors. The synthesis of a crystalline salt of the sodium anion Na − was reported in 1974. Chemical Properties of Non-metals with Reactions - Embibe \ ( {\rm {CC}} { {\rm {l}}_4}\) does not conduct electricity as it contains a covalent bond and has no ions for bonding. Study Chemical Properties of Metals Reaction with Hydrogen Non-metals react with hydrogen to form covalent hydrides. The sharing of electrons forms the non-metal hydrides. How do I derive metallic aluminum without electricity? I'm laying the foundation for a project called 21st Century steampunk, where I figure out what the world would look like if electricity was never discovered. I'm wondering if it's possible to derive usable metallic aluminum from naturally occurring substances without using the Hall-Héroult process.
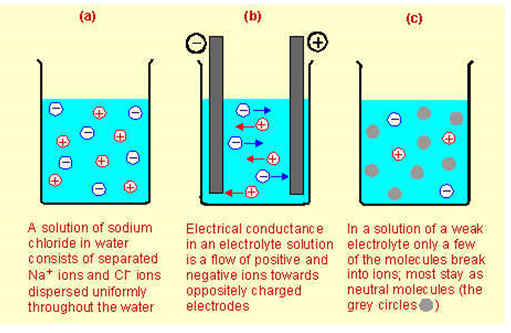
7 Reasons Why Potassium Is Good for You - Sportskeeda Role of Potassium in the Human Body Potassium is the third most abundant mineral in the human body and is essential to the working of the body. When it dissolves in water, it produces positively... potassium | Definition, Properties, & Reactions | Britannica Potassium metal is soft and white with a silvery lustre, has a low melting point, and is a good conductor of heat and electricity. Potassium imparts a lavender colour to a flame, and its vapour is green. It is the seventh most abundant element in Earth's crust, constituting 2.6 percent of its mass. Potash mine at Esterhazy, Sask. Best Electrolyte Supplements in Poland - 2022 The amount of dosage of the best electrolyte supplements in Poland to be taken depends upon the age of the individual. Let's see the details of dosage. For people of age below 50 years, the recommended dosages are - Sodium 1,500mg, Potassium 4,700mg, Calcium 1,000mg, Magnesium 320mg for men and 420mg for women, Chloride 2,300mg. Why Are Ionic Solids Poor Conductors Of Electricity Ionic compounds conduct electricity when molten (liquid) or in aqueous solution (dissolved in water) because their ions are free to move from place to place. Ionic compounds cannot conduct electricity when solid as their ions are held in fixed positions and cannot move.
Noble Gases - Periodic Table | ChemTalk These monatomic gases also conduct electricity and can fluoresce. Because of their nonreactivity, one organic chemist even referred to them as the "lazy elements." However, Neil Bartlett showed us another side to these elements in 1962 by discovering a compound of xenon. Techiescientist - Page 49 of 51 - Science is fun! Does Potassium Conduct Electricity? Potassium is a chemical element that lies in the first group of the periodic table. It is classified as an alkali metal. It is the seventh most abundant element present … Read More. Does Salt Water Evaporate? Acids, Bases and Salts as Electrolytes - Embibe Exams Because they all produce ions when dissolved in water, they can be separated into acids, bases, and salts. Because the positive and negative ions, known as cations and anions, are mobile in these solutions, they conduct electricity. When strong electrolytes are dissolved, they completely ionise, and no neutral molecules form in the solution. can potatoes make electricity? - Test Food Kitchen Yes, potatoes can make electricity. In fact, a small, 2-volt potato battery can power a digital clock or other electronics. Potatoes are made up of cells and each cell contains a small amount of energy.
Homemade Electrolyte Powder (easy natural DIY sports drink!) It does a better job, it's way cheaper, ... Potassium maintains normal cell function, intracellular fluid volume and trans-membrane electrochemical gradients. An absence of potassium can cause constipation, heart palpitations, muscle cramps, increased blood pressure and kidney stones. ... Electrolytes conduct electricity when combined with ...
What does an base produce? - Vivu.tv They are called superbases, and it is impossible to keep them in aqueous solutions because they are stronger bases than the hydroxide ion. Therefore, they deprotonate hydronium ion, conjugate acid of water. Do bases produce electricity?
What's The Deal With Electrolytes? - Cronometer Blog Electrolytes are a group of minerals which include calcium, magnesium, potassium and sodium. What Do They Do? These minerals transmit nerve signals in your body through a form of electro-chemical energy. That means they have a positive or negative charge when dissolved in water and can conduct electricity in that state.
The Best Electrolyte Supplements For Fasting, Athletes, And More - BarBend Electrolytes are minerals that conduct electricity in water, and in doing so, help the body maintain a host of functions — including muscle contraction, and regulating blood pressure.
Hydronephrosis Treatment, Symptoms, Causes, Diagnosis - MedicineNet Electrolytes are substances that become ions in solution and acquire the capacity to conduct electricity. The balance of the electrolytes in our bodies is essential for normal function of our cells and our organs. Common electrolytes include sodium, potassium, chloride, and bicarbonate.
Potassium - Wikipedia In the periodic table, potassium is one of the alkali metals, all of which have a single valence electron in the outer electron shell, that is easily removed to create an ion with a positive charge - a cation, that combines with anions to form salts. Potassium in nature occurs only in ionic salts.
5 Potassium-rich Foods You Need To Add To your Diet - Doctor.ndtv.com Our cells contain almost 98 per cent of the potassium in our body. The majority of this is present in our muscle tissue, and the remaining (20%) is found in our liver, bones, and red blood cells. Potassium also works as an electrolyte in our bodies. Electrolytes conduct electricity in the body and help manage crucial functions in the body.
Liquids Conduct Electricity - Chemical effect of electric current A solution of sodium hydroxide, potassium hydroxide or base in water conduct electricity. Copper sulphate, common salt also conduct electricity. There are differences in the conduction of electricity by solids and liquids.
Which compounds in sports drinks are electrolytes? A substance that dissociates into ions in solution or in the melt acquires the capacity to conduct electricity. Sodium, potassium, chloride, calcium, magnesium, and phosphate in a liquid phase are examples of electrolytes. What is the best drink to replace electrolytes? 8 Healthy Drinks Rich in Electrolytes. Coconut water.
5 Potassium-rich Foods You Need To Add To your Diet - 02XL Our cells contain almost 98 per cent of the potassium in our body. The majority of this is present in our muscle tissue, and the remaining (20%) is found in our liver, bones, and red blood cells. Potassium also works as an electrolyte in our bodies. Electrolytes conduct electricity in the body and help manage crucial functions in the body.
Electrolyte Challenge: Orange Juice Vs. Sports Drink - Science Buddies The ions in the solution give it the capacity to conduct electricity. Electrolytes, such as sodium and potassium, are present in sweat. Chloride, calcium, and phosphate ions are also electrolytes. The proper concentration of electrolytes in your blood is essential to your health.
Difference Between Strong Base and Weak Base They are also good electrolytes. Strong bases are a good conductor of electricity because the ability to conduct electricity depends on the number of ions present and bases have a concentration of ions.. Some of the examples of strong bases are- Sodium hydroxide (NaOH), Potassium hydroxide (KOH), Lithium hydroxide (LiOH), Calcium hydroxide (Ca(OH)2), Rubidium hydroxide (RbOH), Barium hydroxide ...
Did ammonia conduct electricity? - askingforanswer.com They consist of a metal Mg (magnesium) and a polyatomic ion (sulfate) as well as potassium metal and sulfate ion. Both of these compounds can conduct electricity. Is calcium hydroxide a good conductor of electricity? Calcium Hydroxide can conduct electricity only when it is in aqueous solution state or molten state.







0 Response to "42 does potassium conduct electricity"
Post a Comment