42 ice cream lab chemistry answers
Ice Cream Lab: chemistry, cream, en, endothermic, exothermic ... This makes the ice colder than it was before, which is why the ice cream freezes. 1/2 cup of milk1/2 cup of whipping cream1/4 cup of sugar1/4 tsp of vanilla1. Mix ingrediens above in a plastic bag and seal it shut2. in a large bag mix >~2 cups of ice >1/2 cup of saltplace small plastic bag inside3. rub the bag until the mixture solidifies 4. The CHEMISTRY of Ice Cream - scienceteacherprogram.org Here's the recipe to refer to: 1. In small bag, place 1 c. milk, 1/3 c. sugar, and ½ tsp. vanilla. the small bag. 2. In the large bag, get ice (fill just under halfway) and a scoop of salt. Put the small bag inside the larger bag, seal it up. 3. SHAKE. Minimum of 10 minutes. 4. Check for solidification. (while you answer the analysis questions.)
Ice Cream Chemistry - Home This website discovers and researches why flavor scientist use Rock salt in the ice cream process.We discovered that the main reason Rock salt is used is due to the freezing point depression it causes. We developed a procedure to observe which of four salts, would get to the closest degree at which the perfect consistency of ice cream is created.

Ice cream lab chemistry answers
Solved ACTIVITY REPORT: ICE CREAM MAKING Colligative | Chegg.com Chemistry questions and answers. ACTIVITY REPORT: ICE CREAM MAKING Colligative properties are properties of solutions that depend on the number of solute particles dissolved in the solution. There are four colligative properties of solutions, namely, vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure. Ice Cream Lab And Application Questions Answers School of Pastry & Baking Arts | Career Programs | NY Campus | ICEFormer Borden ice cream plant near Unity Park becoming multi …Frozen Hard Ice Cream Machine Lcd Display 110v Commercial - Vevor USGitHub - schwabe/ics-openvpn: OpenVPN for AndroidRapid NMR Fat Analyzer - CEM CorporationBasic Inferential Statistics // Purdue Writing LabTarget ... PDF Chemistry of Ice-Cream Making: Lowering the Freezing Point Preparation of Ice Bath 1 . Fill the Styrofoam cup (or 400 mL beaker) 3/4 full with ice. 2 . Cover the ice with 1/4 to 1/2 inches of table salt. 3 . Stir this ice-salt mixture with a spoon or stirring rod. 4 . Use the thermometer to check the temperature of the ice-salt mixture. It should be at least −10° C. 5 .
Ice cream lab chemistry answers. The CHEMISTRY of Ice Cream Worksheet must be approved before you begin. Analysis: 1. What state of matter was the milk when you began? 2. What state of matter was the milk when you were done? 3. In order to change the phase of the milk, what had to be removed? 4. What happened to the heat energy that left the milk? 5. Why was salt added to the ice? 6. Ice, Cream... and Chemistry - American Chemical Society A gallon (3.8 liters) of ice cream must weigh at least 4.5 pounds, making the minimum density 0.54 gram per milliliter. Better brands have higher densities—up to 0.9 grams per milliliter. The next time you visit a grocery store, compare cheaper and more expensive brands by holding a carton in each hand—you should be able to notice a difference. Classroom Resources | Coffee Creamer Ice Cream | AACT Fill your bottle or bag ½ full with ice. Pour the ¼ cup of sodium chloride (salt) into your bottle. Fill the bottle with ice. Add water about ½ full. Measure the temperature of the mixture in your bottle. Record the temperature in your data table below. Place the lid on your container. Shake your bottle for 1 minute to mix everything together. PDF Make Ice Cream in a Baggie - teachnlearnchem.com Countertop Chemistry Experiment 15 Ice Cream Adding a solute to a solvent lowers the freezing point of that solvent. This change in freezing point is referred to as a colligative property. In this experiment, you will use the lowered freezing point of water to chill another mixture (ice cream) to the solid state. Materials Subsitutions
The Sci Guys: Science at Home - SE1 - EP10: Melting Points: Ice Cream ... Welcome to the tenth episode of The Sci Guys. In this episode we will be investigating an experiment involving making ice cream in a bag with melting points and how we can lower the temperature of... Ice Cream Lab : 9 Steps (with Pictures) - Instructables Make up some ice cream to discuss solutions, mixtures, saturation points, melting point, freezing point and physical changes. The ice cream lab can be used at several grade levels. I use it in the fourth grade to discuss physical changes. I also used this activity in 5th, 6th, and 8th grade over my 25 years teaching. DOCX Home - Scott County Schools LAB . OBJECTIVES: To use both the laws of thermodynamics and properties of solutions to aid in freezing an ice cream mixture. ... C in the shade and to cool off, you are eating an ice cream cone. As you sit there you wonder just how ice cream is made. One area of chemistry that helps to explain the making of ice cream is thermodynamics. There ... PDF Ice Cream Lab & Application Questions - THIRTEEN Ice Cream Lab Answers for Analysis & Application Questions 1. ... (The ice, the milk, the salt, the air in the baggie, the heat from your hands. The milk is the
PDF THERMODYNAMICS & ICE CREAM - Driftwood Lib the ice cream could become. The ice cream mixture would still be a liquid. To freeze the ice cream mixture, it is necessary to use "colder" ice. Again, properties of solutions provide the answers. A salt-ice mixture has a lower freezing point than pure ice, so it acts as "colder" ice. The more salt added to the ice, the lower the ... Ice Cream in a Bag | Chemical Education Xchange ½ cup whole milk, 1 Tbsp Sugar, ¼ tsp. vanilla 2. Fill a quart size freezer bag ½ full of ice. Record the temperature of the ice. Also record the room temperature. 3. Add ½ cup of ice cream salt to the ice. Place the pint size freezer bag into the quart size freezer bag and seal the bag carefully (remove as much air as you can). 4. PDF ICE,Cream Chemistry and - American Chemical Society a lot of air to ice cream is that it tends to melt more quickly than ice cream with less air. The amount of air also has a huge effect on the density of the ice cream. A gallon (3.8 liters) of ice cream must weigh at least 4.5 pounds, making the minimum density 0.54 gram per milliliter (or 540 grams per liter). better brands PDF Interdisciplinary Making Ice Cream In A Bag - CDÉACF 7. The mixture will freeze into ice cream in about 5 to 9 minutes. 8. When the ice cream feels frozen, remove the inner bags and rinse the outside of the bag with water to remove any of the salt mixture. 9. Use your spoon and eat your homemade ice cream right out of the bag! 10. Dispose of the plastic bags and spoon in the garbage. 11. Reverse ...
ice-cream-lab.doc - I SCREAM, YOU SCREAM, WE ALL SCREAM FOR ICE CREAM ... Fill the large bag ½ full with ice. 4. Pour 1 cup of rock salt into the ice bag and seal the bag. 5. Gently squeeze the ice salt mixture being careful not to open the bag. 6. Continue to squeeze and rotate the ice cream bag until the ice cream reaches the desired consistency - about 6-8 minutes. 7.
PDF Lab 24: Colligative Properties and Ice Cream - Catholic Texts Lab 24: Colligative Properties and Ice Cream 5. The freezing point of a substance is a physical property. How did this experiment demonstrate that "freezing point" of a substance is a colligative property? 6. Imagine you performed this experiment and accidently added one mole of sugar instead of one mole of sodium chloride to the bag of ice a.
DOC Lab: Colligative Properties - Mrs. Murphy's Chemistry Class (BE SURE TO RECORD ANY OBSERVATIONS!) 1. Get a Ziploc bag. Fill it half full of ice. 2. Using the paper cup add about 4 tablespoons rock salt to the bag. 3. Place the creamer container inside the bag. 4. Shake the bag well for ten minutes. 5. While shaking, measure the temperature of the ice mixture every minute for the 10 minutes.
PDF COLLIGATIVE PROPERTIES ICE CREAM LAB - Molelady 2 cups and 2 spoons (use spoon to measure teaspoon amount) thermometer Procedure: 1. Record the temperature of the ice. 2. Place "A" ingredients in quart-size Ziplock baggie. Be sure to remove as much air as possible before closing baggie. 3. Place "B" ingredients in gallon-size zip-lock baggie along with filled closed quart-size baggie. 4.
Ice Cream Lab Sheet - Google Docs Procedure: (Follow the directions carefully). 1. Add 1/4 cup sugar, 1/2 cup milk, 1/2 cup whipping cream, and 1/4 teaspoon vanilla to the quart (small) Ziploc bag. Seal the bag securely. 2. Put 2-3...
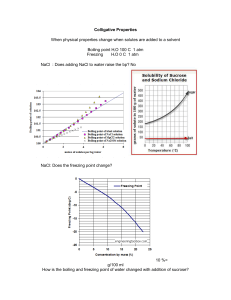
Lab 2 - Ice Cream Chemistry - Alliance Chemistry Ice Cream Chemistry Part V In this lab you will have sodium chloride (NaCl), sucrose (table sugar), water, and a heat source. You will be measuring the change in boiling point and using this change to determine the molar mass of each of these solutions. This lab procedure should be broken up into 3 parts:
DOC Colligative Properties: Making Ice Cream: - Georgetown ISD 1. In the 1-quart Ziploc bag place the milk, sugar, flavoring, and seal the bag carefully. Place this bag in the larger (1-gallon) Ziploc bag. 2. In the larger Ziploc bag put in ice (fill the bag about half full) and ½ cup of rock salt. Carefully seal the larger Ziploc bag. Place Ziploc bag into a trash bag. 3.
PDF Ice Cream Lab - Santa Ana Unified School District / Overview securely or else you will have SALTY ice cream!! 6. GENTLY knead the bags so that the ice cream solution is thoroughly mixed as it freezes. 7. Knead the bags for 10 minutes or until the solution reaches "Ice Cream" consistency. It's going to get cold so be careful! 8. When finished, open the gallon bag and remove the quart bag. 9.
experiment 3: chemistry of ice cream Data Sheet Table | Chegg.com Transcribed image text: Post-Lab Questions 1. Shortly after you added the salt to the ice cubes, was the temperature above or below the freezing temperature for water? 2. Why does saltwater freeze in the arctic, but salt is used to melt ice? 3. Heat energy is needed to change phase from a solid to a liquid.
Homemade Ice Cream in a Bag Science Experiment - Study.com Steps. 1. In each mixing bowl, whisk together one cup of half and half, 2 tablespoons sugar and 1/2 teaspoon vanilla extract. Pour the contents of each bowl into a pint-size resealable plastic bag ...
PDF Chemistry of Ice-Cream Making: Lowering the Freezing Point Preparation of Ice Bath 1 . Fill the Styrofoam cup (or 400 mL beaker) 3/4 full with ice. 2 . Cover the ice with 1/4 to 1/2 inches of table salt. 3 . Stir this ice-salt mixture with a spoon or stirring rod. 4 . Use the thermometer to check the temperature of the ice-salt mixture. It should be at least −10° C. 5 .
Ice Cream Lab And Application Questions Answers School of Pastry & Baking Arts | Career Programs | NY Campus | ICEFormer Borden ice cream plant near Unity Park becoming multi …Frozen Hard Ice Cream Machine Lcd Display 110v Commercial - Vevor USGitHub - schwabe/ics-openvpn: OpenVPN for AndroidRapid NMR Fat Analyzer - CEM CorporationBasic Inferential Statistics // Purdue Writing LabTarget ...
Solved ACTIVITY REPORT: ICE CREAM MAKING Colligative | Chegg.com Chemistry questions and answers. ACTIVITY REPORT: ICE CREAM MAKING Colligative properties are properties of solutions that depend on the number of solute particles dissolved in the solution. There are four colligative properties of solutions, namely, vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.










0 Response to "42 ice cream lab chemistry answers"
Post a Comment